Just One Centimetre Deeper: Place Cells and the Brain's Cartography
This article originally appeared, in a shorter form, in the ‘Neural Musings’ column on Neu-Reality. I have since rewritten and expanded it considerably.
‘I sometimes feel, in reviewing the evidence on the localisation of the memory trace, that the necessary conclusion is that learning just is not possible.’ Karl Lashley wrote this in 1950, after thirty-three years of cutting into the brains of rats and asking where memory lives. He had trained animals to run mazes, then systematically lesioned piece after piece of cortex, expecting that one cut would erase the route. None did. The amount of tissue removed predicted the impairment; the location did not. His conclusion was bleak and elegant: memory is everywhere and therefore, in any surgically useful sense, nowhere. ‘It is difficult to conceive of a mechanism which can satisfy the conditions set for it. Nevertheless, in spite of such evidence against it, learning does sometimes occur.’
The remark was half joke, half despair. It became one of the most quoted sentences in neuroscience, and it was wrong — but wrong in a way that mattered. Lashley looked only at the cortex. Had he gone one centimetre deeper, he would have hit a curved, layered structure tucked beneath the temporal lobe whose name, given by the Italian anatomist Giulio Cesare Aranzi in 1587, means seahorse: the hippocampus. Inside it was the map he had been looking for. But it would take another two decades, a shy Quaker’s heresy, and an accidental electrode before anyone found it.
Edward Tolman arrived at the University of California, Berkeley in 1918 and stayed for the rest of his life. He was a pacifist’s son from Massachusetts, shy, self-deprecating, and, by his own telling, an accidental psychologist. He had gone to MIT ‘not because I wanted to be an engineer, but because I had been good in mathematics and science in high school and because of family pressure’. He drifted into psychology at Harvard, got his PhD, and was promptly fired from his first faculty post at Northwestern for publishing in a pacifist student magazine during wartime. Berkeley took him in. He ran rats through mazes. He thought about what the rats were doing.
This was more controversial than it sounds. The dominant framework was Clark Hull’s stimulus-response mechanics at Yale, which treated the brain as a telephone switchboard: stimulus in, response out, nothing interesting in between. Tolman proposed something scandalous: that rats were thinking. That when they ran a maze, they were not merely chaining together habits but building an internal representation of the environment: a map. His research, one account noted, ‘evoked from his behaviourist colleagues an agonised search for hidden errors and the fear that he was undermining the development of “objective psychology true and narrow”’. The division had a geographic dimension. As another later observed: ‘Place-learning organisms, guided by cognitive maps in their heads, successfully negotiated obstacle courses at Berkeley, while their response-learning counterparts, propelled by habits and drives, performed similar feats at Yale.’
The evidence came from mazes, but the best of them told a story. In the latent learning study with Charles Honzik in 1930, Tolman ran three groups of food-deprived rats through a fourteen-unit T-maze. The first group always found food at the end. The second never did. The third got nothing for ten days — then, on day eleven, food appeared. Overnight, their error rate plummeted to match the always-rewarded rats, as though they had been learning the maze silently all along but had no reason to show it. The implication for pure stimulus-response theory was devastating: learning could happen without reinforcement.
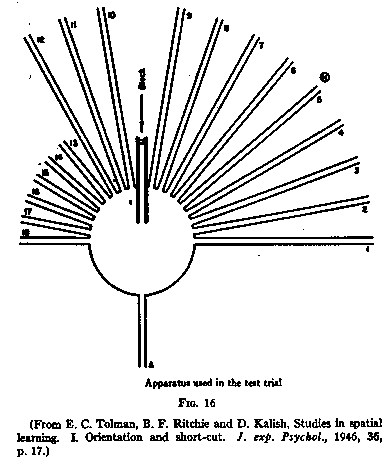
Then came the sunburst maze. Tolman, Ritchie, and Kalish trained rats to run from a start box, across an open circular platform, through a single walled alley to a food box at the far end. After four nights, the apparatus was transformed: the original path was blocked, and a fan of eighteen radiating arms replaced it. The rats returned to the platform, found themselves blocked, explored the radiating arms — and preferentially chose the one pointing directly toward where the food had been. They were not retracing a route. They were consulting a representation.
The central nervous system is far more like a map control room than it is like an old-fashioned telephone exchange.
Tolman called this representation a ‘cognitive map’, and his 1948 paper in the Psychological Review, ‘Cognitive Maps in Rats and Men’, remains one of the most extraordinary documents in the history of psychology. It argued that learning consists not in stimulus-response connections but in building up neural structures that ‘function like cognitive maps’, and that these maps vary from ‘narrow strip’ varieties (a single rigid path from position to goal) to ‘broad comprehensive’ ones (a wide representation of surrounding space that supports flexible rerouting).
Then the paper makes a leap that no one expected. In the final pages, Tolman scales up from mazes to politics. He identified four conditions that produce narrow strip maps (brain damage, inadequate environmental cues, excessive repetition of one path, and ‘the presence of too strongly motivational or of too strongly frustrating conditions’) and argued, with characteristic self-deprecation (‘a rat psychologist’s ratiocinations offered free’), that the same narrowing operates in human societies. Over-motivated, frustrated people develop narrow cognitive maps and displace their aggression onto outgroups. ‘The poor Southern whites, who take it out on the Negroes, are displacing their aggressions from the landlords.’ Americans criticising Russians, departments sniping at rival departments: all ‘nothing more than such irrational displacements’. His closing is a moral imperative framed in the language of rat psychology: ‘We dare not let ourselves or others become so over-emotional, so hungry, so ill-clad, so over-motivated that only narrow strip-maps will be developed.’
A year later, when Berkeley’s Board of Regents imposed a loyalty oath on faculty, it was Tolman, the shy Quaker who was too diffident to seek faculty leadership, who stood before the Academic Senate and read a public letter to President Sproul: ‘The issue I am concerned with involves not communists but liberals.’ He was fired. He sued. The resulting case, Tolman v. Underhill, reached the California Supreme Court and struck down the oath. In 1963, four years after Tolman’s death, Clark Kerr named Berkeley’s new psychology building Tolman Hall.
Tolman proposed the cognitive map but had no way to see inside the brain. Hull’s camp mounted effective counterarguments. B. F. Skinner published ‘Are Theories of Learning Necessary?’ in 1950, arguing that psychologists should describe behaviour rather than hypothesise about mental states. The cognitive map was shelved. The fifties and sixties were Hull’s and Skinner’s decades. Only with the cognitive revolution of the late 1960s did the intellectual climate begin to shift; by then, someone working in a basement in London had already found the cells.
John O’Keefe was born in Harlem in 1939, to Irish immigrant parents who arrived just before the Depression. His mother’s passage to America had been funded by an uncle and redeemed by seven years’ indentured labour. His father spent most of his career as a night-shift bus mechanic, fixing vehicles ‘which couldn’t be fixed by the daytime mechanics’. Neither parent had completed elementary school.
O’Keefe won a scholarship to Regis, the Jesuit school in Manhattan, but felt like an outsider and ‘never got to grips with Latin and Greek’. He left, worked, then studied aeronautical engineering in the evenings at NYU while commuting over a hundred miles daily. During electives he became drawn to philosophy and concluded that ‘many of the perennial problems in philosophy might be solvable through brain research’. When he decided to quit his job at Grumman, the company counter-offered by inviting him to work on the Lunar Expedition Module for the Apollo programme. He turned it down. He calls it, in his Nobel autobiography, ‘a path not taken…’
He drove a taxi at night through New York to support himself through college. At City College he ranged freely (filmmaking, literature, physics, psychology, philosophy) until a dean called him in, pointed out he had enough credits for multiple degrees, and asked him to ‘please choose one and get out’. He chose physiological psychology. At McGill, under Ronald Melzack, he found an extraordinary environment shaped by Donald Hebb, Brenda Milner, Peter Milner, Wilder Penfield, and Herbert Jasper. Recording from the amygdala, he discovered highly specific cells (‘mouse detectors, specific food detectors, and bird song detectors’) which led him to formulate what he called his ‘first law of the nervous system: the silent cells are the important ones’.
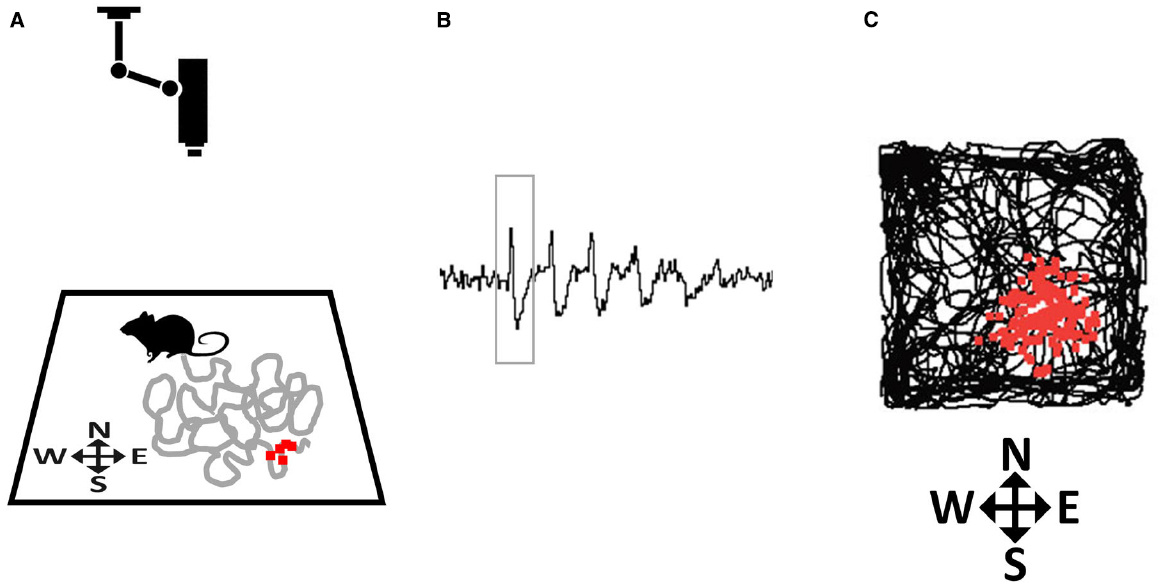
He moved to University College London in 1967, as a postdoc in Patrick Wall’s lab. The hippocampal work began, as he put it, ‘from an experiment which went astray’. He was trying to record from the somatosensory thalamus when his electrode strayed into the hippocampus. The first cell that he found was a theta-correlated interneuron. He decided then and there to leave the somatosensory system. He was drawn by the hippocampus’s many silent cells: ‘here was a brain region chock-a-block full of them.’
He was not looking for spatial coding. He was looking for memory cells, inspired by Brenda Milner’s decades of work on patient H.M., Henry Molaison, the man who lost both hippocampi to surgery in 1953 and woke into a world where every five minutes erased the last. O’Keefe thought: ‘If I’m going to really go for the home run, as it were, I should go and look and find the memory cells in this part of the brain.’
With Jonathan Dostrovsky, an MSc student passing through the lab, he recorded from seventy-six neurons during every behaviour he could observe: eating, drinking, grooming, exploring, lever-pressing. Over months he grew suspicious that certain cells’ activity related not to what or why the animal acted but had ‘something to do with where it was doing it’. Then, as he later recalled: ‘On one electrifying day I realised with a flash of insight that the cells were responding to the animal’s location.’
They ruled out sound by silencing fans, smell by rotating the platform. The cells were not sensory detectors. They were building an inner map. O’Keefe recognised that they might represent ‘the neural site of Tolman’s cognitive map’. He experienced, he said, ‘a prolonged euphoria of the classical Archimedean type’.
The first paper was initially rejected by Brain Research, then accepted after revisions. O’Keefe ‘confidently sat back and waited for the chorus of approval’. Instead: ‘There was a deafening silence, with the exception of a small number of isolated voices.’ The phenomenon was hard to communicate in print. As one later reviewer observed: ‘It is very difficult to convey in words the compelling nature of place-field activity.’ The early figures were marginally convincing, and there was no quantitative analysis. The field’s resistance ran deeper than methodological objection. Lashley had spent thirty-three years showing that you could not pin function to a place in the cortex. O’Keefe was claiming exactly the opposite, that specific cells in a specific brain region encode specific locations. The titan’s shadow was long.
In 1976, O’Keefe formally named these neurons ‘place cells’. (Strictly speaking, he used ‘place unit’, since extracellular recording lacks single-cell resolution; the informal term stuck.) The spatial representation constructed by place cells could, in principle, support all the navigational functions Tolman had postulated. Consider a maze: if, after repeated trials, a rat has formed a cognitive representation of the layout, place cells could ‘pave’ a cognitive pathway connecting entrance to exit. But cells blanketing the environment are not sufficient on their own. The brain also needs to know which pairs of cells represent adjacent locations and in what order they are linked. O’Keefe found the cognitive map in the hippocampus. How rats actually use it to navigate was the next question.
In 1978, O’Keefe and the psychologist Lynn Nadel published The Hippocampus as a Cognitive Map, a book that opens, startlingly for a neuroscience monograph, with a sixty-page philosophical survey covering Kant, Leibniz, Newton, Poincaré, William James, Piaget, and Gibson. O’Keefe endorsed Kant’s position that spatial sense is innate and a priori, and asked: ‘Had we found the neural basis for Kant’s a priori spatial faculty of sensibility?’
The book distinguished ‘locale’ navigation (map-based, hippocampal) from ‘taxon’ navigation (route-based, stimulus-response), and reinterpreted virtually every hippocampal lesion study through the lens of spatial deficits, remarkable because, as the authors acknowledged, ‘virtually none of the original studies interpret the results in terms of space’. They described their interpretive style as ‘chiaroscuro’: they ‘avoided shades of grey’. An Oxford University Press reviewer (reportedly the foremost expert on animal behaviour) delivered a long, blisteringly negative assessment. Rather than abandon the project, O’Keefe and Nadel spent six years rewriting it entirely.
The result was cited by many and read by few. The neuroscientist John Kubie admitted that ‘decades ago, on first reading, I got lost about ten pages in’ to the philosophy chapter. Used copies reportedly cost around $700; O’Keefe eventually made it freely available online. Kubie compared the discovery of place cells deep in the brain, far from sensory inputs or motor outputs, to discovering the answer ‘42’ to the ultimate question in The Hitchhiker’s Guide to the Galaxy — ‘without knowing the question’.
It took until the mid-1980s, when Robert Muller and John Kubie introduced computerised data acquisition and produced the first clean colour-coded rate maps, for the field to broadly accept place cells. O’Keefe and Speakman’s 1987 experiment was also decisive: they showed that after the removal of cues, place fields remained stable — and predicted which arm the rat would choose. The cells, as the authors wrote, ‘fired when the animal evidently thought he was in the place-field’. Evidently thought. The language had changed.
The next question was temporal: not just where but when. O’Keefe turned to the firing patterns of place cells in relation to the hippocampal local field potential, and in particular to a distinctive oscillation first mapped by Green and Arduini in 1954: theta, a rhythm of six to ten cycles per second that appears when a rat moves. (The name is slightly wrong: rat theta averages around eight hertz, technically outside the original theta band, but the label had already stuck by the time anyone measured properly.)
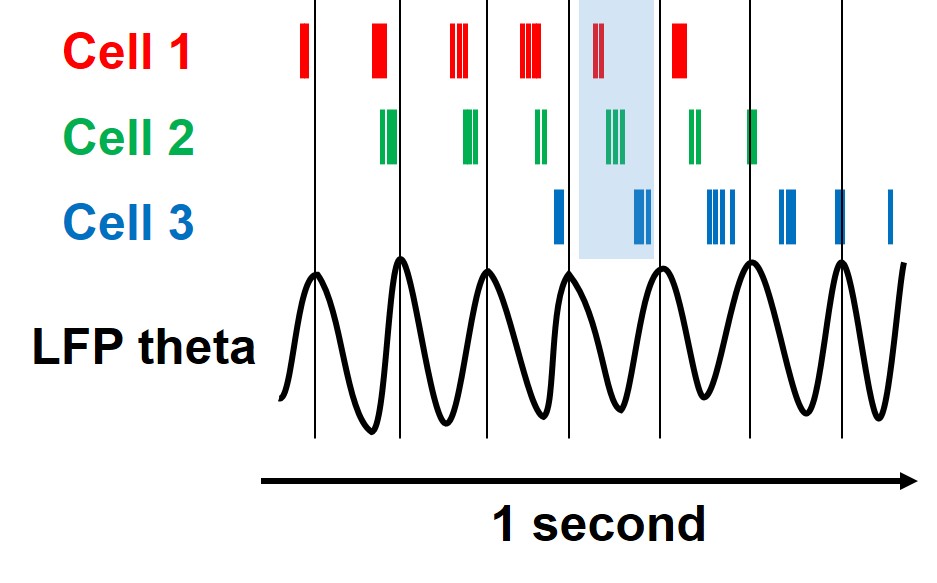
In 1993, O’Keefe and Michael Recce discovered phase precession — a temporal code riding on the theta rhythm. As a rat enters a place cell’s firing field, the cell fires at a particular phase of the ongoing theta cycle — say, near the peak. As the rat moves through the field, each successive burst of spikes occurs slightly earlier in the theta cycle. By the time the rat exits, the cell is firing near the trough. The spike timing has ‘precessed’ through nearly a full 360 degrees of the cycle.
The mechanism O’Keefe and Recce proposed, the ‘detuned oscillator’ model, has a beautiful simplicity. Imagine two metronomes ticking at almost but not quite the same tempo. Because the place cell’s intrinsic oscillation is slightly faster than the background theta rhythm, its firing gradually drifts earlier relative to each cycle, like one metronome’s clicks slowly overtaking the other’s. The result: firing rate peaks in the centre of the field (where the two oscillations are most in sync), and spike timing encodes position within the field with finer precision than firing rate alone. The hippocampus was not just saying ‘you are here’ through which cells fired. It was encoding where within the field the animal was through when the cell fired relative to the ongoing rhythm. A temporal code layered on top of a rate code.
Phase precession at the single-cell level gives rise to something remarkable at the population level: theta sequences. Within a single theta cycle — roughly 125 milliseconds — place cells with overlapping fields fire in a compressed temporal sequence that recapitulates the order of fields the animal is traversing. The compression ratio is striking: up to ten to one. A trajectory that takes the animal a second or more to physically travel is replayed in the hippocampus within about a hundred milliseconds. This is not merely efficient coding. The compressed intervals bring successive place cells’ spikes within the twenty-millisecond window required for spike-timing-dependent plasticity — the mechanism by which synapses learn sequential structure. The theta sequence is, in effect, a teaching signal: it compresses experience to the timescale at which synapses can learn.
The sequence does not merely record where you have been; it predicts where you will be.
And theta sequences look forward. They represent not just where the animal has been but where it is going. Wikenheiser and Redish showed in 2015 that the forward extent of theta sequences, how far ‘ahead’ of the animal they project, varies with the animal’s goals. On journeys to distant goals, look-ahead extended farther. The hippocampus was not passively recording trajectory. It was projecting it.
This became even more striking in Kay, Chung, Sosa, and colleagues’ 2020 paper in Cell, from Loren Frank’s laboratory at UC San Francisco. They asked what theta sequences do when the animal faces a choice. Recording from rats on a W-shaped maze with a fork in the middle, they found that as the animal approached the choice point, theta sequences did not commit to one path. Instead, within adjacent theta cycles, they alternated — on one cycle representing the left arm, on the next representing the right arm, cycling back and forth at theta frequency, roughly every 125 milliseconds. The hippocampus was rapidly simulating both possible futures, as though deliberating between them.
The cycling was constant and rapid, not restricted to moments of overt hesitation. Even at full running speed, the hippocampus was toggling between possible futures. The alternation was linked to ‘cycle skipping’ at the single-cell level, where individual place cells fire on alternate theta cycles rather than every cycle. Most striking: the decoded sequences did not reliably predict which path the rat would actually choose. The hippocampus was representing both options with roughly equal probability, maintaining what amounts to a parallel simulation rather than committing to a decision. This reframes hippocampal theta sequences as a mechanism for generativity: the capacity to represent hypothetical, not just actual, experience. The cognitive map does not merely reflect the world. It imagines alternatives.
The cognitive map’s coordinate system turned up not in the hippocampus itself but one synapse upstream, in the entorhinal cortex, and the people who found it had started their scientific lives by making a poster pink to tease a professor.
May-Britt and Edvard Moser met as teenagers in western Norway. They reconnected as students at the University of Oslo, married in 1985, and burned, as May-Britt later put it, ‘with eagerness to understand the brain’. They wanted to study neural mechanisms, not just behaviour, but the neurophysiologist Per Andersen — gatekeeper to the neuroscience group — was sceptical of psychologists, and his lab was full. Andersen, as president of the European Neuroscience Association, allowed them to present a poster at a conference in Sweden. May-Britt made it pink, ‘as a way to tease Per a bit’. The neuroscientist Richard Morris walked past, noticed their dorsal-ventral findings, and mentioned the poster during his plenary talk. The Mosers were in.
Their postdoctoral work in O’Keefe’s lab at UCL was, in May-Britt’s words, ‘one of the most learning-rich periods in our lives’. O’Keefe sat with them in the surgery room, showed them how to turn down tetrodes, run recordings, cluster data. They were offered two assistant professorships at NTNU in Trondheim before finishing their postdocs — an offer so unexpected they nearly turned it down. They had planned to stay longer with O’Keefe or go to Arizona, ‘a real neuroscience mecca at the time’. But two jobs and a lab of their own was too good to refuse. For a long stretch in Trondheim it was only the two of them, doing ‘everything from cutting brains to cleaning the rat cages’.
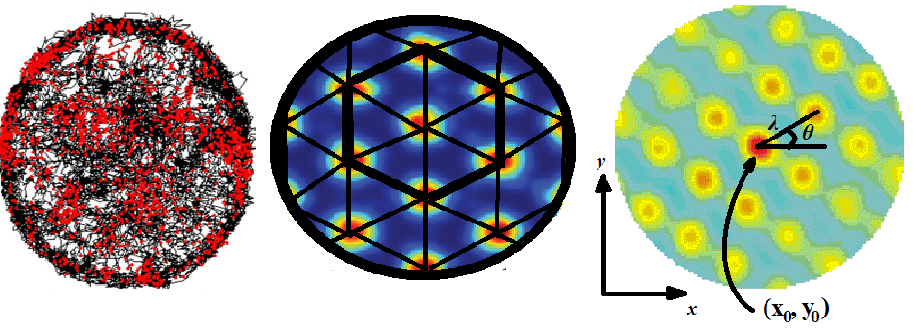
In 2004, recording from the entorhinal cortex (a region almost nobody else was studying, partly because one side lies dangerously close to a large blood chamber), they noticed that neurons fired at multiple locations, with hints of regularity. But the recording box was too small for the periodicity to be visible. The decisive step was simple: they moved the rats into a larger environment. That was when the hexagonal grid appeared.
Edvard described the realisation as ‘a long-drawn-out eureka moment’: not a single flash but a dawning recognition that these cells fired in a perfect, repeating triangular lattice, like the vertices of tiles on a bathroom floor. He compared it to ‘a hole in a Chinese checkerboard’. The pattern was generated entirely by the brain; there was no hexagonal template in the outside world. Grid cells came in different scales, field spacing from about thirty to a hundred and fifty centimetres, and the pattern persisted in darkness, independent of landmarks, speed, or direction, suggesting it depended on path integration: the brain’s dead-reckoning system.
The 2005 paper in Nature by Hafting, Fyhn, Molden, Moser, and Moser was ‘so different from what anyone had expected’. O’Keefe’s own group had looked in the entorhinal cortex for spatial cells and had missed the grid. The discovery completed a picture that had been assembling for fifteen years: alongside place cells in the hippocampus, the navigational system included head direction cells (discovered by Taube, Muller, and Ranck in 1990, firing when the animal’s head points in a specific direction, an internal compass), and border cells (reported by Solstad, Boccara, Kropff, and the Mosers in 2008, firing at environmental boundaries). Place cells say you are here. Grid cells provide the metric: distance and direction. Head direction cells orient the system. Border cells anchor the map to walls and edges.
The Nobel committee, in 2014, called it an inner GPS. O’Keefe received one half of the prize; the Mosers shared the other. On October 6, 2014, May-Britt was in a lab meeting when an unknown number rang. She told the caller: ‘No, I don’t believe you, please can you send me an e-mail so that I can read it because I don’t believe my ears.’ Edvard was on a plane to Munich, working on a manuscript. He was met at the gate by an airport representative with flowers and a car.
The historical arc the prize recognised began long before any of the laureates. Santiago Ramón y Cajal, working with Golgi staining around 1901, had drawn the hippocampal circuit in India ink and graphite (the dentate gyrus, CA3, CA1) and added arrows to indicate the direction of impulse flow, turning a still image into what amounted to a circuit diagram. A teenager trained at a provincial art academy, Cajal had bought his first microscope in 1877 with money saved from service as an army medical officer. His drawings of the hippocampus are still reproduced in contemporary textbooks. From Cajal’s ink arrows to O’Keefe’s place cells to the Mosers’ grid: the same structure, seen first in dead tissue, then in the living brain.
The hippocampus entered human medicine before it entered spatial theory, and it did so through catastrophe. On 25 August 1953, the neurosurgeon William Beecher Scoville removed tissue from both medial temporal lobes of Henry Molaison — a twenty-seven-year-old with debilitating epilepsy — including most of both hippocampi, the amygdalae, and the entorhinal cortex. The seizures abated. But Molaison woke into a condition that no one had anticipated: he could no longer form new memories. If someone introduced themselves, left the room for five minutes, and returned, he had no recollection of the meeting. He described his condition as ‘like waking from a dream… every day is alone in itself’.
Brenda Milner, working with Wilder Penfield at the Montreal Neurological Institute, studied him for roughly thirty years. He was always polite (he would say good morning if you passed him in the waiting room) but he had no idea who you were. He met Milner hundreds of times and never remembered her. Yet when she asked him to trace the outline of a five-pointed star while seeing only its reflection in a mirror, his performance improved steadily over ten trials — and the next day, tested again, he showed clear retention of the skill. He had no memory of ever having performed the task. The hippocampus was essential for declarative memory (facts and events) but procedural memory lived elsewhere.
The 1957 paper by Scoville and Milner became one of the most cited in neuroscience. Patient H.M., as Molaison was known for decades, established the hippocampus as indispensable for converting transient experience into lasting memory. What no one yet understood was why the same structure that held memories also held maps. That connection, between where you are and what you remember, would take another half-century to articulate.

The human evidence came from multiple directions. In 2000, Eleanor Maguire and colleagues at UCL used structural MRI to compare the brains of sixteen licensed London taxi drivers with fifty controls. To earn a licence, drivers must pass ‘The Knowledge’ — three to four years of training memorising roughly 25,000 streets within a ten-kilometre radius of Charing Cross, along with thousands of landmarks and routes. The result: taxi drivers had significantly larger posterior hippocampi, and the volume correlated with years on the job. In a follow-up longitudinal study, Maguire and Katherine Woollett tracked seventy-nine aspiring drivers over four years. Those who passed The Knowledge showed posterior hippocampal growth; those who failed did not. The posterior expansion came at the apparent cost of the anterior hippocampus, suggesting that mastering spatial memory might carry a trade-off.
Then came the direct recordings. In 2003, Arne Ekstrom, Itzhak Fried, and colleagues at UCLA recorded from 317 individual neurons in seven epilepsy patients who had electrodes implanted for surgical planning. While the patients navigated a virtual town using a joystick, 24 per cent of hippocampal neurons fired at specific spatial locations — place cells, directly analogous to those O’Keefe had found in rats thirty-two years earlier. A decade later, Joshua Jacobs and colleagues found grid-like cells in the human entorhinal cortex. The inner GPS was not a rodent specialisation. It was a mammalian inheritance.
The clinical shadow of this system is Alzheimer’s disease, in which the entorhinal cortex and hippocampus are among the first structures to degenerate. Getting lost in familiar environments is often the earliest symptom noticed by families, years before a formal diagnosis. The map frays before memory does, or rather, the fraying of the map is the fraying of memory, because the two were never separate.
The discovery that pulled the hippocampal story beyond space came in 2011, from Howard Eichenbaum’s laboratory at Boston University. While rats waited during a temporal gap in a sequence task, hippocampal neurons fired at successive moments during the delay — each cell ‘preferring’ a particular moment in time, much as place cells prefer particular locations in space. Eichenbaum called them ‘time cells’. Just as place cells remap when the environment changes, time cells retime when temporal parameters are altered. The hippocampus appeared to use the same computational machinery to map both spatial and temporal dimensions of experience.
Time cells were not the only sign that the cognitive map extended beyond geography. ‘Splitter cells’ fire selectively for one of two directions at a fork, encoding not just where the animal is but where it came from and where it is headed. ‘Vector trace cells’ fire when an animal is at a specific position relative to an object and continue firing after the object is removed, a memory of spatial relationship persisting in the absence of its referent. In humans, Tavares and colleagues showed in 2015 that the hippocampus maps dimensions of social space (power and affiliation), not just physical coordinates. And in the monkey hippocampus, Knudsen and Wallis found cells encoding the ‘value’ of different images, how rewarding they were, with activity patterns that closely resembled those seen in rodents during spatial navigation.
These cell types do not necessarily map one-to-one onto individual neurons: the same cell can play different roles in different environments, contexts, or tasks. The hippocampus, it now appears, is not a spatial map with memory as a side effect, nor a memory system that happens to encode space. It is a relational engine, a structure that organises the elements of experience, whatever their nature, into the coherent sequences and frameworks we call episodes, routes, plans, and knowledge. As György Buzsáki and Edvard Moser argued in 2013, the mechanisms of memory and planning may have evolved from mechanisms of navigation in physical space. Episodic memory, mental travel through remembered experience, evolved from dead-reckoning navigation. Semantic memory, knowledge of facts and categories, evolved from landmark-based, map-like navigation. Remembering where you were last Tuesday and finding your way home may be, at the deepest computational level, the same operation.
The cognitive map was never just about mazes.
And during sleep, the map replays. In 1994, Matthew Wilson and Bruce McNaughton showed that place cells which fired together during the day’s exploration exhibited an increased tendency to fire together during subsequent sleep — but not during sleep that preceded the experience. The day’s spatial sequences were being replayed, compressed into sharp-wave ripples: brief, high-frequency bursts representing the most synchronous population events in the mammalian brain. Not all experiences are replayed equally. The hippocampus selects. Yang and colleagues showed in 2024 that ripples occurring during reward consumption on the maze effectively ‘tagged’ those sequences for later replay during sleep. The map consolidates itself by rehearsing what mattered.
From Tolman’s shy heresy in Berkeley, through Lashley’s magnificent failure to find the engram, through O’Keefe’s accidental electrode and the Mosers’ hexagonal lattice, the story of the cognitive map has been one of successive enlargements. Tolman proved the map existed by watching rats choose the right arm. O’Keefe found its neurons. The Mosers found its coordinate grid. Eichenbaum showed it maps time. Others have shown it maps social relationships, abstract values, hypothetical futures. The structure Aranzi named after a seahorse in 1587 turns out to be less a map of physical space than a map of the relationships between things, where things include places, moments, people, rewards, dangers, and ideas.
Tolman, in his 1952 autobiography, credited everyone but himself: ‘First of all most of the credit, if it be credit, should go to all the students whose ideas I have shamefully… adopted and exploited… and ended up by believing to be my own.’ He died in 1959, twelve years before O’Keefe found the cells that proved him right. He never knew. But the paper that ends with a plea against narrow strip maps — against the cognitive impoverishment that comes from hunger, fear, and frustration — reads differently now. The hippocampus really does build broad comprehensive maps. And the conditions that narrow them — stress, deprivation, threat — really do shrink the hippocampus, as decades of research on chronic stress and cortisol have since confirmed. Tolman’s metaphor was not a metaphor. It was a prediction, dressed in the self-deprecating language of a man who thought he was merely speculating about rats.
The principal sources for the historical narrative are O’Keefe’s Nobel and Kavli autobiographies; May-Britt Moser’s Nobel autobiography and the Mosers’ Nobel interview; David Redish’s Beyond the Cognitive Map (MIT Press, 1999); the historical review in Best and White, ‘Hippocampal cellular activity: A brief history of space’ (PNAS, 1998); and Bhatt et al. (2016). On the Tolman–Hull rivalry: Tolman’s ‘Cognitive Maps in Rats and Men’ (1948). On H.M.: the Scoville and Milner paper (1957) and the Journal of Neuroscience retrospective. On London taxi drivers: Maguire et al. (2000) and Woollett and Maguire (2011). On grid cells: Hafting et al. (Nature, 2005). On theta sequences at choice points: Kay et al. (Cell, 2020). On time cells: MacDonald et al. (Neuron, 2011). On sleep replay: Wilson and McNaughton (Science, 1994). On the hippocampus as relational engine: Buzsáki and Moser (Nature Neuroscience, 2013). O’Keefe and Nadel’s The Hippocampus as a Cognitive Map is freely available online.
Originally published in Chinese as 「位置细胞:大脑探索世界的导盲杖」 on Neu-Reality.