The brain does not have a single mode. The same cells, the same synapses, the same hippocampus can compute one thing or another depending on the chemical state they happen to be in. I want to understand how neuromodulation, dopamine in particular, reconfigures neural circuits to enable learning and memory.
how I got here
I read Psychological and Behavioural Sciences at Magdalene College, Cambridge. Within the first year I knew I wanted to be closer to the biology: I was already sitting in on neuroscience and zoology lectures at the Downing Site, drawn to the machinery underneath the behaviour. I transferred to Natural Sciences in my second year.
Cambridge gave me three things I did not expect. The first was Paul Brakefield, who supervised my Part IA Evolution and Behaviour paper and taught me how to sit with a problem, how to think through it slowly, as a biologist, rather than reaching for an answer. The second was David Parker, whose lectures on motor circuits and synaptic plasticity were among the best I have ever attended: precise, passionate, and always reaching for the deeper principle. The third was the freedom to follow what I found interesting, including philosophy, which became a lasting passion and changed how I think about explanation in science.
Fisher Lab, UCL · summer 2018
My first time in a real laboratory was at the Queen Square Institute of Neurology, in Elizabeth Fisher’s group. I was a Genetics Society summer student, working on cathepsin B activity in mouse models of Down syndrome–associated Alzheimer’s pathology: western blots, protein activity assays, plaque counting. Paige Mumford, the Masters student at the time, took me under her wing; Claudia Manzoni and Karen Cleverley helped me find my footing; Frances Wiseman taught me how to think about experimental design. Fisher herself showed me what it means to think carefully about disease and genetics. That work eventually became part of a published paper, and I presented my cathepsin B results at the Genetics Society Summer School in Edinburgh that August.

I did not stay in disease research. The summer had confirmed that I wanted a life in science, but it also pointed me toward a different scale: not proteins and plaques, but cells and circuits. A paper I had encountered in a Cambridge lecture kept pulling me back. Bi and Poo’s 1998 work on spike-timing-dependent plasticity showed that the order and timing of spikes could write the rules of learning, and that felt like the right level of question. It pointed me toward synapses, plasticity, and eventually toward neuromodulation: the layer that decides whether a synapse can change in the first place.
Paulsen Lab, Cambridge · 2019–2020

I asked Ole Paulsen for a summer position in his lab and was taken in by Tanja Fuchsberger, who taught me two things: how to do whole-cell patch-clamp electrophysiology, and how to think about dopamine and plasticity. At the time, Bittner et al.’s work on behavioural-timescale synaptic plasticity from Jeff Magee’s lab was a major reference point. Tanja was asking whether dopamine might act as a threshold-reducer, whether biology uses it to let weak associations, protocols that would normally fail, induce long-term potentiation. I tested that idea for my undergraduate thesis and found that dopamine did reduce the stimulation intensity required for LTP induction. I stayed in the lab after the summer and completed my thesis there. It was awarded the Bundy Scholarship.

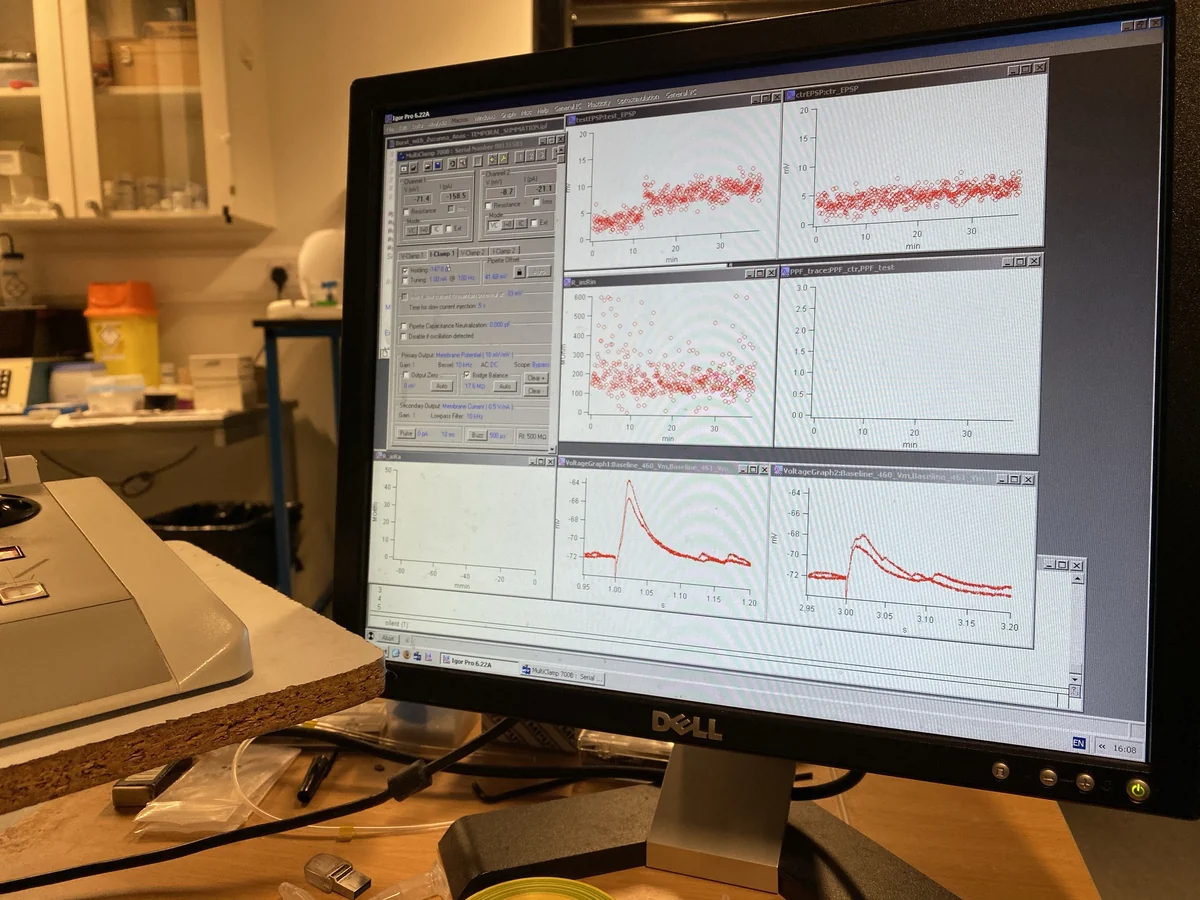
That year crystallised two convictions. First, that dopamine is not a reward signal alone; it is a gain controller, a gate, something that changes what circuits can do. Second, that I wanted to move from single synapses to populations: to ask not just whether one synapse strengthens, but how a neuromodulatory signal reshapes what an ensemble of neurons computes.
between Cambridge and Florida
I graduated in July 2020 and spent the next year at Scientific American China in Beijing before starting my PhD. I arrived at the Max Planck Florida Institute for Neuroscience in May 2021.

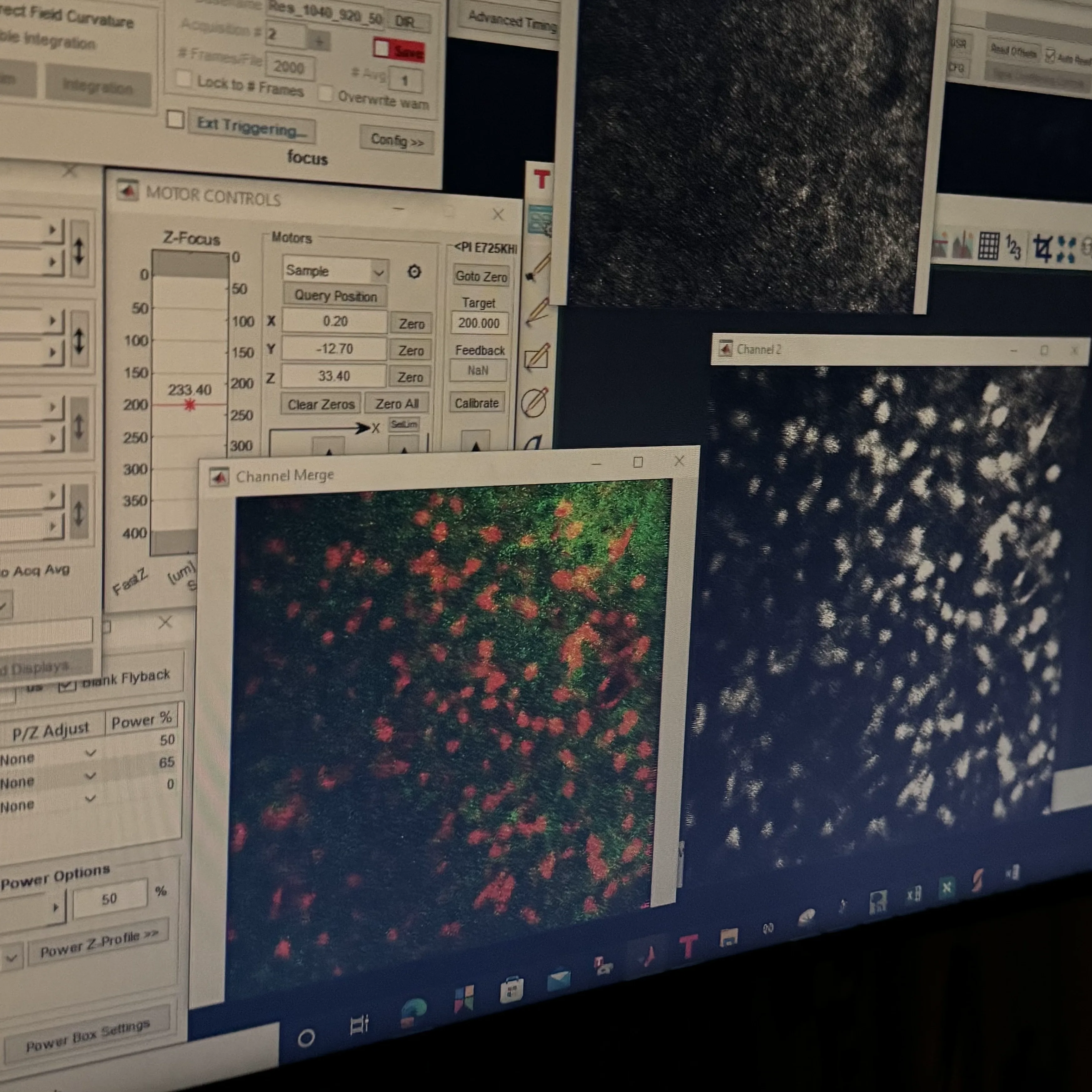
Everything was new. I had come from patch-clamp, which is intimate and slow: one cell, one pipette, one afternoon. Now there were surgeries to learn, stereotaxic injections, craniotomies. Raphael Heldman and Kori-Anne Citrin, the senior PhD students in the lab, taught me the procedures. The computational side came more naturally, though it was no less steep. I taught myself population analysis techniques and sought out expert friends for the parts I could not work out alone. Gradually the questions that had been forming since the Paulsen Lab, about how neuromodulation reshapes what a population of neurons computes, became questions I could actually test.
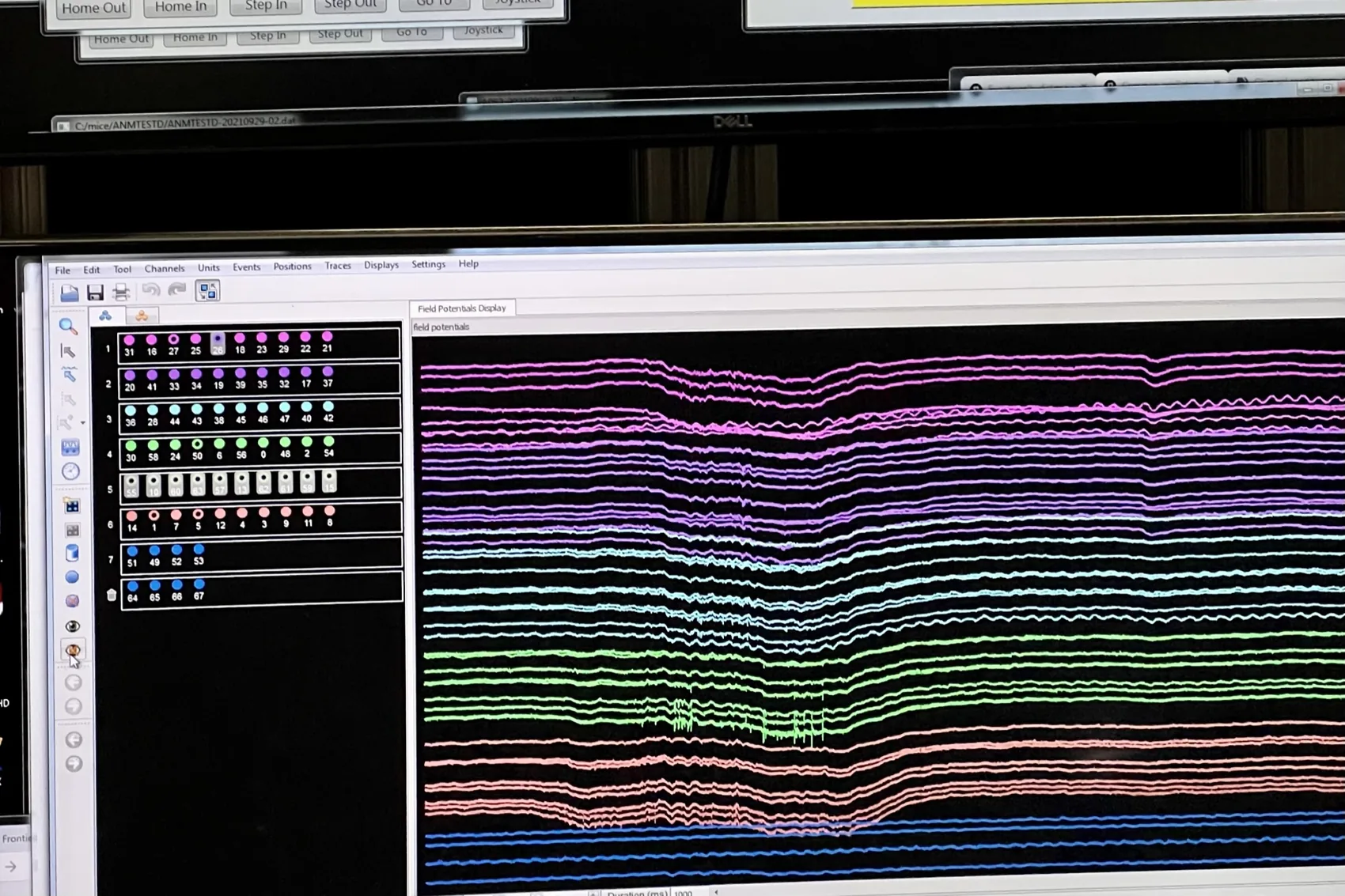
The first time I saw multiple neurons firing together on a screen, through a Neuronexus silicon probe, marked a shift. In patch-clamp one hears a single cell at a time with extraordinary intimacy. Now there was a population, and with it came a different set of questions and a pull toward computational neuroscience: PCA, LDA, UMAP, the whole vocabulary of dimensionality reduction that lets one ask what an ensemble is doing rather than what a single neuron prefers.
current project
Rapid locus coeruleus dopamine shapes CA1 dynamics for time/distance estimation
Manuscript in preparation. I am defending soon.
This is my dissertation work and manuscript-in-preparation; the summary below describes our current evidence and interpretation.
A mouse running through a cue-free virtual corridor has to estimate when the hidden reward zone is near. There are no landmarks, so the animal integrates self-motion: how long it has been running, how far it has travelled, and when to begin licking for reward. My PhD asks how the brain puts that computation into the right state, and the answer points to an unexpected source of dopamine.
what we found
LC activity marks the start of estimation. A major subset of catecholaminergic locus coeruleus (LC) neurons bursts at trial-start run onset. The response is not explained by running speed alone, predicts first-lick timing on single trials, and optogenetic amplification delays reward-seeking without reducing running or reward rate. That single-trial specificity is one of the parts I find most striking: pushing a single moment of LC activity shifts when the animal begins its decision.
The LC signal reaches CA1 as dopamine. Local CA1 pharmacology points to D1-like dopamine receptors rather than alpha-1 or beta adrenergic receptors as the relevant pathway for task performance. Two-photon imaging with dLight, a genetically encoded dopamine sensor developed by Patriarchi, Tian, and colleagues, shows that LC activation and natural run onset both produce fast dopamine signals in CA1. The LC-to-hippocampus dopamine pathway is relatively new (Takeuchi et al. 2016 was among the first widely recognised demonstrations) and much of what the field knows about local dopamine dynamics comes from VTA studies. Seeing it from the LC side is why the project still feels fresh to me.
The dopamine signal is local and short-lived. Around LC axons in CA1, dLight responses are strongest near the axon pixels, fade over micrometre distances, and decay over the next one to two seconds. That is the surprising scale of the result: not a millisecond synapse, and not a slow global bath, but something in between.
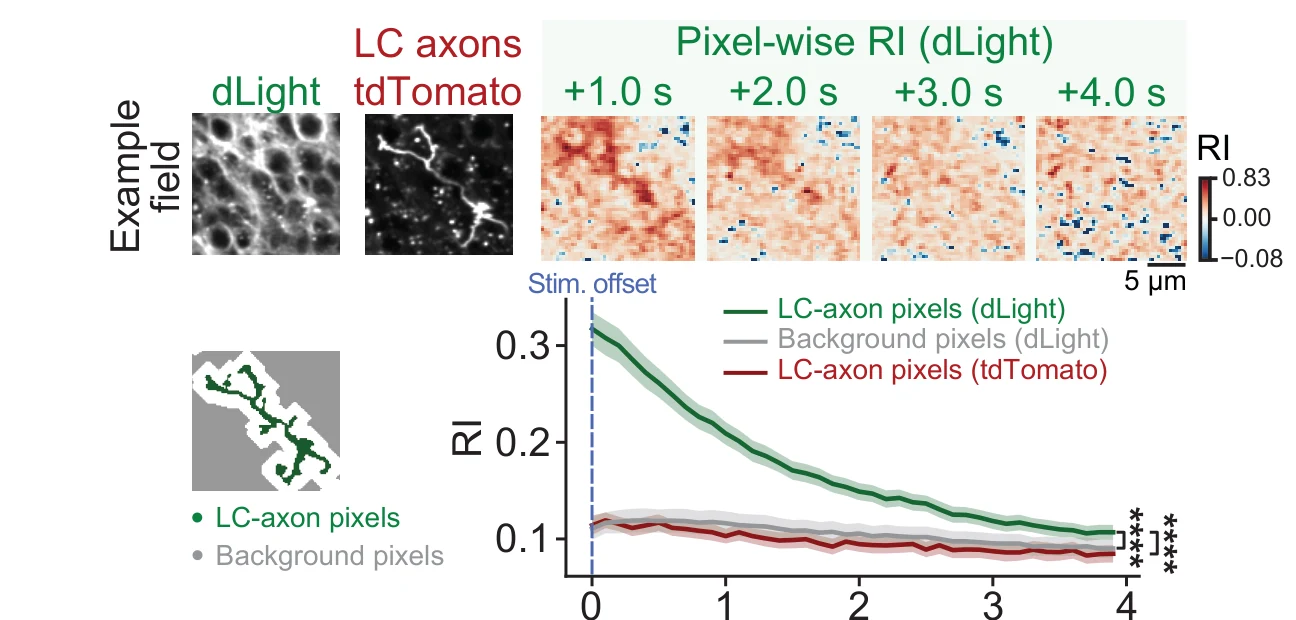
Local dopamine is linked to CA1 population state. CA1 neurons with run-onset dLight signals are enriched for PyrUp dynamics — a population pattern linked to the start of goal-directed running — and D1-like receptor blockade reduces PyrUp recruitment. The behavioural shift, dopamine signal, and CA1 population change all meet on the same timescale.
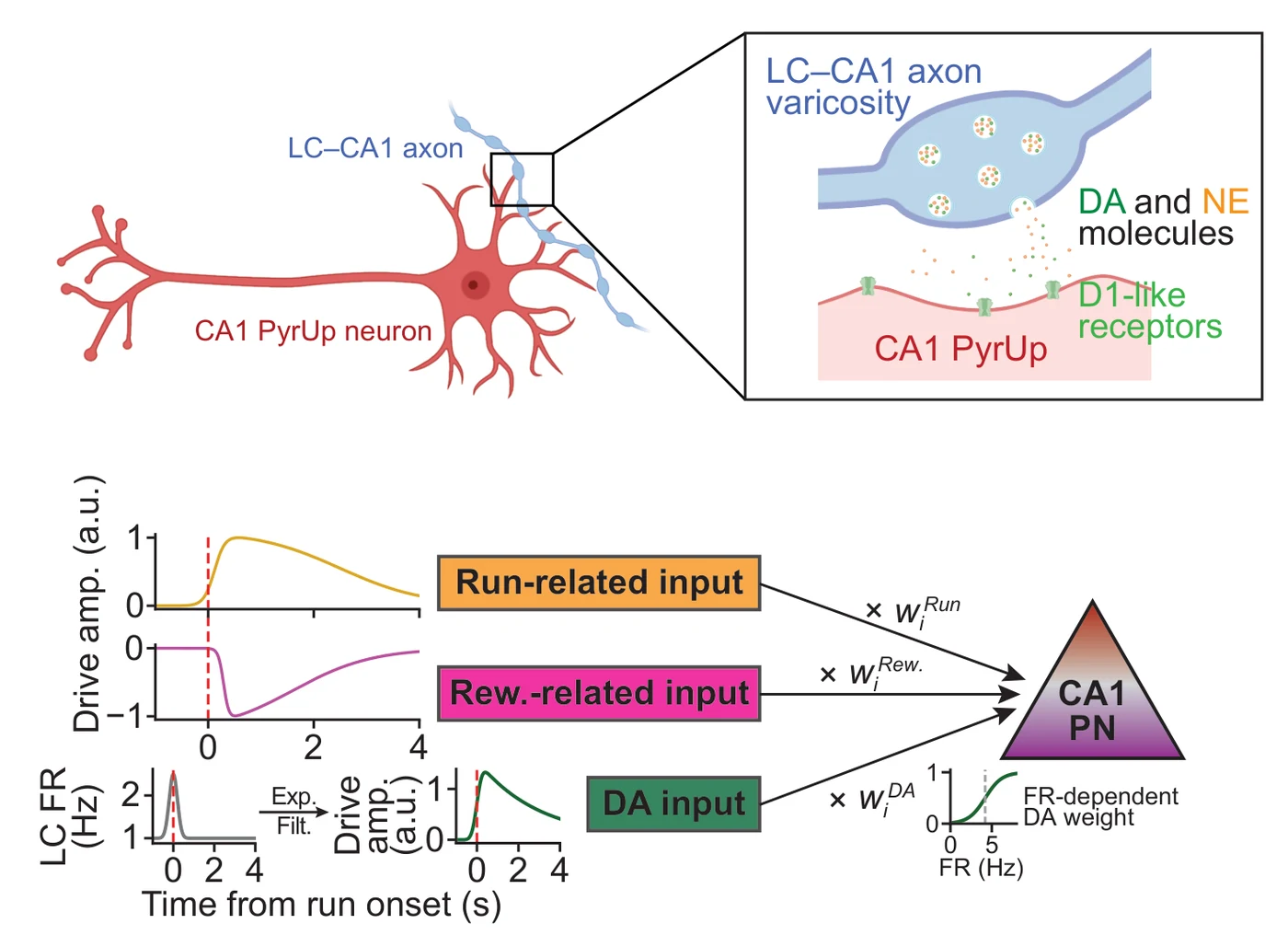
A compact model reproduces the selectivity. The model uses three inputs to a synthetic CA1 principal-neuron population: run-related input, reward-related input, and a phasic LC signal transformed into dopamine drive. The run and reward terms follow Raphael Heldman’s Nature Communications and bioRxiv models of CA1 principal-neuron dynamics; the new term is the LC-to-dopamine drive.
why it matters
Classical neuromodulation is often treated as diffuse, global, and slow. This project shows an intermediate mode: seconds-timescale, micrometre-scale dopamine that prepares a hippocampal computation at the moment a decision begins to unfold. A circuit can change state quickly without rewriting the whole network.
That connects back to what I first saw in the Paulsen Lab, that dopamine is not just a reward signal but a gain controller. The difference is the scale at which it operates. In the Paulsen work the question was whether dopamine lowers the threshold for a single synapse to change. Here the question is whether a phasic dopamine signal, delivered locally and lasting only seconds, can bias what an entire population of neurons does next. The answer, it seems, is yes.
methods
The project combines head-fixed virtual-reality navigation, LC and CA1 electrophysiology, optogenetics, local CA1 pharmacology, two-photon dopamine and calcium imaging, Python analysis, and compact computational modelling.
outputs

Manuscript in preparation
Luo D, Cao J, Heldman R, Tian L, Wang Y. Rapid locus coeruleus dopamine shapes CA1 dynamics for time/distance estimation.
Selected presentations
- Locus coeruleus dopamine rapidly modulates CA1 dynamics to sharpen goal-directed navigation. COSYNE 2026, selected poster.
- Locus coeruleus dopaminergic modulation of CA1 shapes behavioural timescale dynamics for time integration. Lake Conference, Seattle, 2025.
- An LC-CA1 circuit for initiating path integration. Sunposium, West Palm Beach, 2025.
- Locus coeruleus regulates hippocampus-dependent integration at single-trial level. Memory Formation Forum, Max Planck Florida Institute for Neuroscience, 2024.
publications and code
Wu Y, Mumford P, Noy S, Cleverley K, Mrzyglod A, Luo D et al. Cathepsin B abundance, activity and microglial localisation in Alzheimer’s disease-Down syndrome and early onset Alzheimer’s disease: the role of elevated cystatin B. Acta Neuropathologica Communications 11, 132 (2023). doi:10.1186/s40478-023-01632-8
Research code and cleaned analysis material will be linked through GitHub and ORCID as the manuscript moves toward publication.
Last updated May 2026